How to Calculate the Standard Molar Enthalpy
An application of Hesss law allows us to use standard heats of formation to indirectly calculate the heat of reaction for any reaction that occurs at standard conditions. About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy Safety How YouTube works Test new features Press Copyright Contact us Creators.

5 1 Standard Enthalpy Changes Of Formation And Combustion Youtube
Enthalpy of combustion of benzene.

. The molar mass of a compound can be calculated by adding the standard atomic masses in gmol. Calculate moles n of fuel used. C If heat is lost to the surroundings instead of being transferred only into the aluminum can and.
286 k J m o l 1. Calculate moles n of fuel used. Calculate the molar heat of combustion.
The Molar Heat Capacity formula is defined as the amount of energy required to raise the temperature of one mole of a substance by one degree. Therefore a better version of this equation is. H 2 g 1 2 O 2 g H 2 O l.
The enthalpy change for this reaction is -5762 kJmol at 25 C. Calculate the molar heat of combustion of ethanol molar enthalpy of combustion of ethanol. Its units in the SI system are Jmol K and is represented as c m Q n T or Molar Heat Capacity Heat Number of Moles Change in Temperature.
B Use tabulated standard enthalpies of formation to calculate a theoretical value for the molar. The molar mass is the mass of a given chemical element or chemical compound g divided by the amount of substance mol. You can calculate changes in enthalpy using the simple formula.
Moles mass molar mass. -5762 kJmolFrom this the standard enthalpy change H is obtained by division with the amount of substance in moles involved. Nov 18 2019 Click here to get an answer to your question Compute the molar enthalpy of combustion of glucose C6 H12O6.
1054 107J 1 kJ 103J 1054 104 kJ. 1mole NaOH 6322 J 600 106moles NaOH 1054 107 J. In this equation the delta symbol or means a change in Normally you would hold the pressure as a constant value.
In molar heat of neutralization problems n CV where. Heat released or absorbed mass specific heat capacity change in temperature. A Calculate the molar enthalpy of combustion of benzene using the experimental evidence.
Δ c H. Calculate the standard enthalpy of formation Key Takeaways Key Points The standard state of a material is a reference point for the materials thermodynamic state properties. An enthalpy change that occurs specifically under standard conditions is called the standard enthalpy or heat of reaction and is given the symbol.
Calculate energy required to change temperature of water. But for constant pressure the standard enthalpy change simply refers to transferred heat. Therefore you can say that the enthalpy of dissolution or molar enthalpy of dissolution for sodium hydroxide is.
303 calculate the heat energy change from a measured temperature change using the expression Q mcΔT. Standard enthalpy of combustion is defined as the enthalpy change when one mole of a compound is completely burnt in oxygen with all the reactants and products in their standard state under standard conditions 298K and 1 bar pressure. C6 H12O6 s 6 O2.
Molar enthalpy D Hn. H U PV. Q m cg Tfinal - Tinitial q m cg ΔT.
304 calculate the molar enthalpy change ΔH from the heat energy change Q. H H products H reactants Definition of Enthalpy The precise definition of enthalpy H is the sum of the internal energy U plus the product of pressure P and volume V. Finally convert this to kilojoules.
Steps to calculate the molar enthalpy of combustion of ethanol using these experimental results. Heat is the form of energy that is transferred between systems or objects with. Calculation of Molar Enthalpy of Solution molar heat of solution 6 Step 1.
ΔΗ combustion KJ Substance ΔΗ kJmole -2777 00 CHOH1 02g CO2g H2Og -3935 -2418. H U PV. C concentration in M molesL.
Calculate the standard molar enthalpy for the complete combustion of liquid ethanol C2H5OH using the standard enthalpies of formation of the reactants and products. Up to 24 cash back How to calculate molar enthalpy of formation The standard enthalpy of formation refers to the enthalpy change when one mole of a compound is formed from its elements. 305 Triple only draw and explain energy level diagrams.
Moles mass molar mass Calculate energy required to change temperature of water. As the acid and the base are fully dissociated and neither the cation B nor the anion A are involved in the neutralization reaction. N number of moles of reactant.
So we convert the carefully measured mass in to moles by dividing by molar mass. ΔH diss 11 104kJ mol1. To calculate the enthalpy of solution heat of solution using experimental data.
Therefore a standard enthalpy change is. V volume in litres. Calculate the heat released or absorbed in joules when the solute dissolves in the solvent.

Enthalpy Of Formation Video Khan Academy

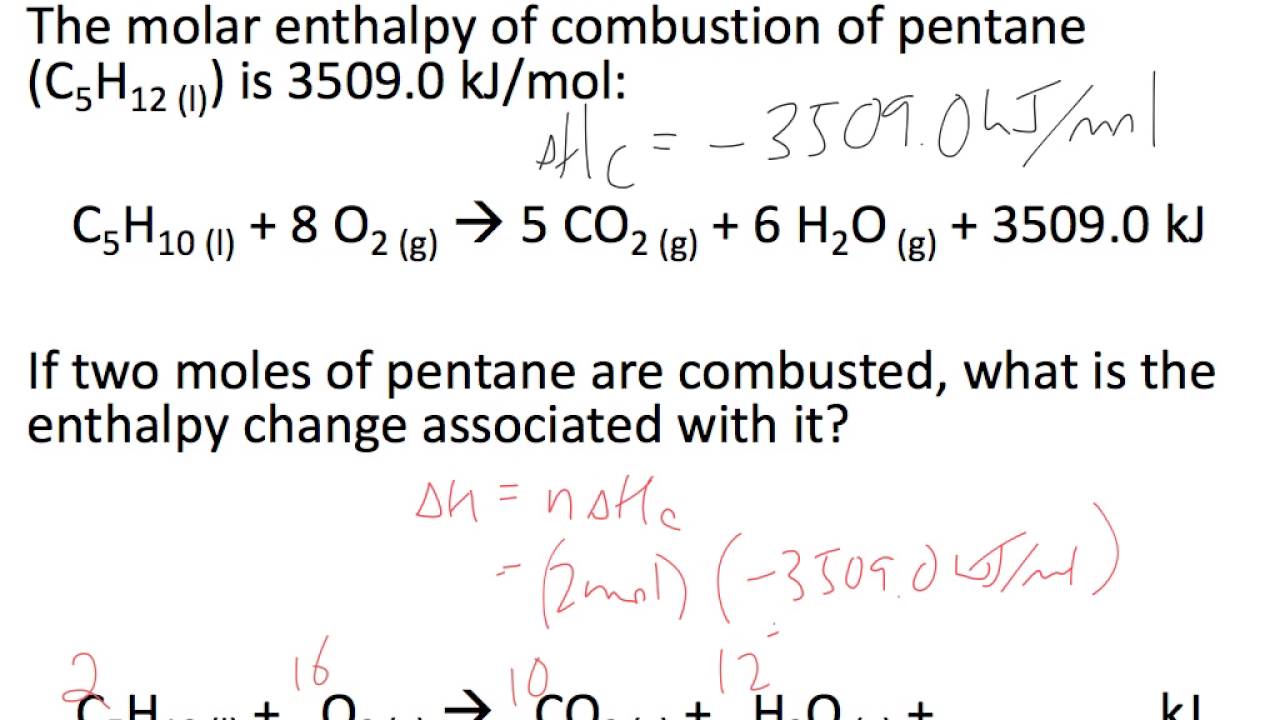
No comments for "How to Calculate the Standard Molar Enthalpy"
Post a Comment